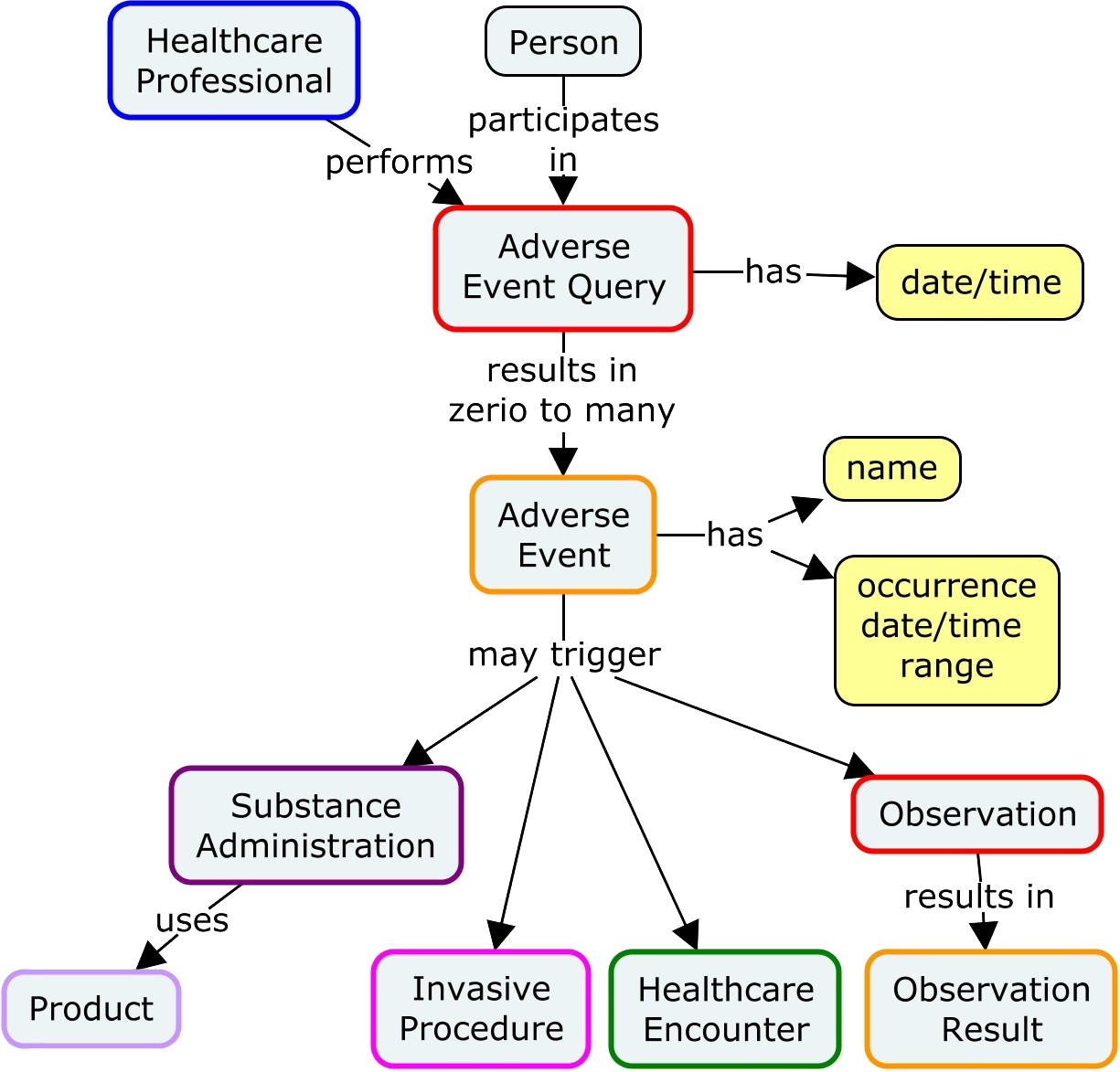
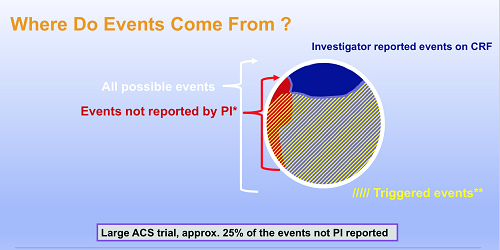
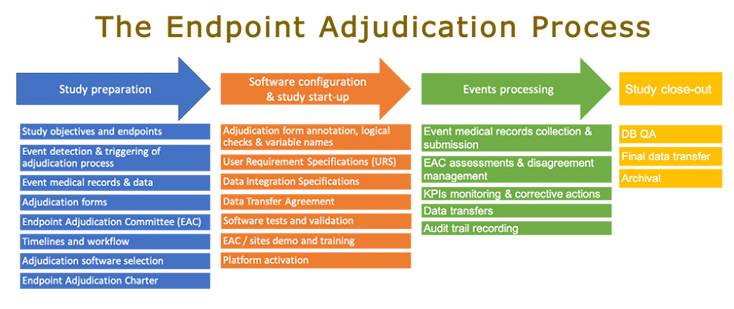
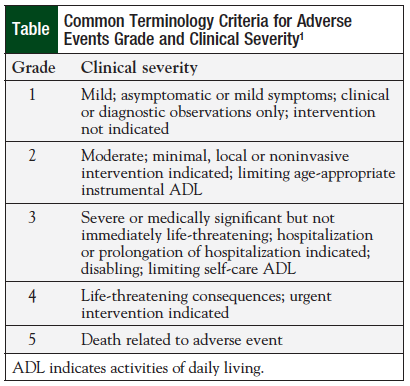
Clinical events classification (CEC) in clinical trials: Report on the current landscape and future directions — proceedings from the CEC Summit 2018 - ScienceDirect
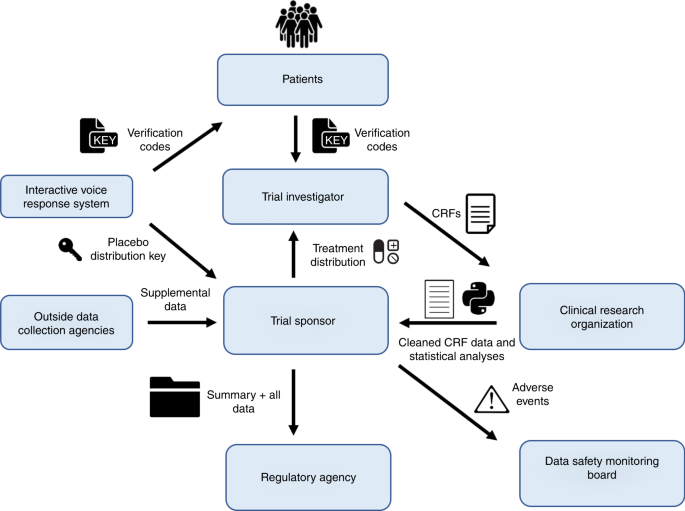
Prototype of running clinical trials in an untrustworthy environment using blockchain | Nature Communications
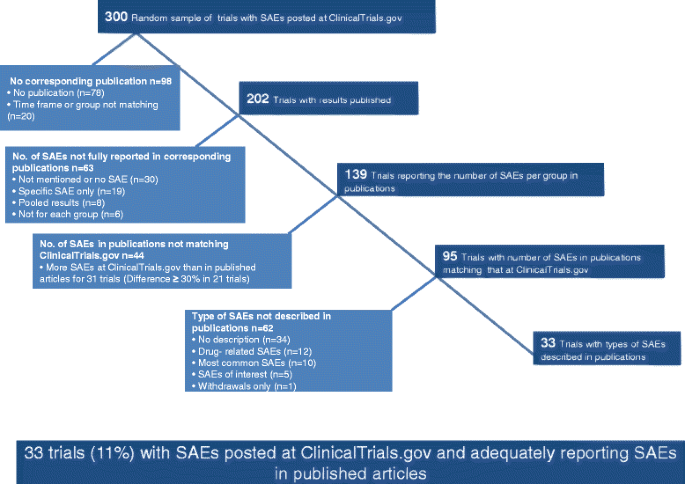
Comparison of serious adverse events posted at ClinicalTrials.gov and published in corresponding journal articles | BMC Medicine | Full Text
Timeline of clinical events and therapeutic interventions, listed as... | Download Scientific Diagram
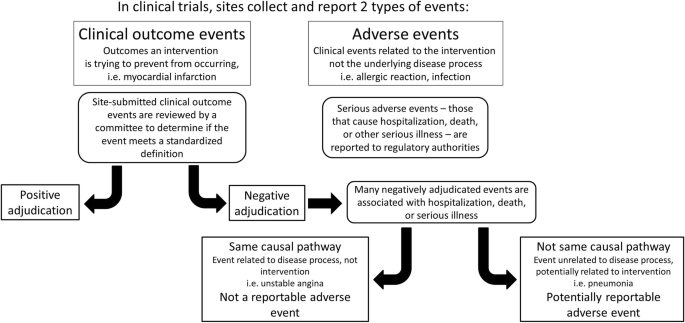
Methods for safety and endpoint ascertainment: identification of adverse events through scrutiny of negatively adjudicated events | Trials | Full Text

Reproducibility of clinical events adjudications in a trial of venous thromboembolism prevention - Girard - 2017 - Journal of Thrombosis and Haemostasis - Wiley Online Library

Predicting severe clinical events by learning about life-saving actions and outcomes using distant supervision - ScienceDirect